|
9/1/2023 0 Comments Entropy change equation 
The Clapeyron relation gives the slope of the tangents to this curve. (c) If a system undergoes a reversible process. Insert this into the definition of entropy. (b) If a system undergoes a reversible change, the entropy of the universe increases. On a pressure– temperature ( P– T) diagram, for any phase change the line separating the two phases is known as the coexistence curve. Equation of state for ideal gas is PV equal to RT, so pressure P is RT over V. Go, for the change using the following equation. James Thomson and William Thomson confirmed the relation experimentally in 1849-50, and it was historically important as a very early successful application of theoretical thermodynamics. From Free Energies of Formation From Enthalpy Changes and Entropy Changes From Equilibrium Constants. Its relevance to meteorology and climatology is the increase of the water-holding capacity of the atmosphere by about 7% for every 1 ☌ (1.8 ☏) rise in temperature. 
It's named after Rudolf Clausius and Benoît Paul Émile Clapeyron. From Equation 4. The Clausius–Clapeyron relation, in chemical thermodynamics specifies the temperature dependence of pressure, most importantly vapor pressure, at a discontinuous phase transition between two phases of matter of a single constituent. Calculate a change in entropy for an irreversible process of a system and contrast with the change in entropy of the universe Explain the third law of thermodynamics We have seen how entropy is related to heat exchange at a particular temperature. The equation for the change of entropy (delta S) of a system or object is the energy transferred to or from the object (Q), measured in Joules, divided by the average temperature of the object (T. The following formula calculates the change in entropy: S (q/T)rev (H/T)rev. 2 ) We can think of Equation ( 5. 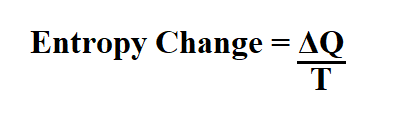
"Clapeyron equation" and "Clapeyron's equation" redirect here. Using the equation of state for an ideal gas (), we can write the entropy change as an expression with only exact differentials: ( 5. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
